Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
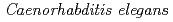
鈴木 芳代; 坂下 哲哉; 小林 泰彦; 辻 敏夫*
no journal, ,
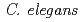 typically approaches NaCl.
typically approaches NaCl. 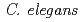 modifies its movement response to NaCl from attraction to avoidance following exposure to NaCl in the absence of food for several hours. The behavioral plasticity in response to NaCl can be explained as the changes over time of both the response-characteristics of each neuron and the degree of neurotransmission. Although the involvement of some neurons in learning has been determined from molecular experiments, whether the change of neurotransmission corresponding to the behavioral changes extends to the whole nervous system or only to a limited part of the nervous system is not known. Therefore, to understand the behavioral changes induced by learning at the neuronal network level, we propose an approach in which the neuronal network is modeled, and the changes of neurotransmission corresponding to learning are estimated. Our results revealed that neurotransmission on multiple synapse connections differed prior to and after chemotaxis learning. This partially corresponded to the experimental findings of previous studies. In addition, our results suggested the involvement in chemotaxis learning of some synapse connections between interneurons, whose roles for learning have never been known. We will discuss the possibility of our computational approach to provide novel information that cannot be obtained using conventional experimental techniques.
modifies its movement response to NaCl from attraction to avoidance following exposure to NaCl in the absence of food for several hours. The behavioral plasticity in response to NaCl can be explained as the changes over time of both the response-characteristics of each neuron and the degree of neurotransmission. Although the involvement of some neurons in learning has been determined from molecular experiments, whether the change of neurotransmission corresponding to the behavioral changes extends to the whole nervous system or only to a limited part of the nervous system is not known. Therefore, to understand the behavioral changes induced by learning at the neuronal network level, we propose an approach in which the neuronal network is modeled, and the changes of neurotransmission corresponding to learning are estimated. Our results revealed that neurotransmission on multiple synapse connections differed prior to and after chemotaxis learning. This partially corresponded to the experimental findings of previous studies. In addition, our results suggested the involvement in chemotaxis learning of some synapse connections between interneurons, whose roles for learning have never been known. We will discuss the possibility of our computational approach to provide novel information that cannot be obtained using conventional experimental techniques.
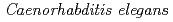
服部 佑哉; 鈴木 芳代; 曽 智*; 小林 泰彦; 辻 敏夫*
no journal, ,
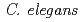 shows some rhythmic movements including the pharyngeal pumping motion for chewing and swallowing. Biological signals can be measured using the electropharyngeogram (EPG), which represents the electrophysiological responses of the pharyngeal muscle cells in a lump, and there is evidence that the pumping rhythms are generated by the pharyngeal muscle cells and controlled by pharyngeal neurons. In addition, we recently reported that the pumping rhythms temporarily change after ionizing irradiation. Thus, the pumping motion in
shows some rhythmic movements including the pharyngeal pumping motion for chewing and swallowing. Biological signals can be measured using the electropharyngeogram (EPG), which represents the electrophysiological responses of the pharyngeal muscle cells in a lump, and there is evidence that the pumping rhythms are generated by the pharyngeal muscle cells and controlled by pharyngeal neurons. In addition, we recently reported that the pumping rhythms temporarily change after ionizing irradiation. Thus, the pumping motion in 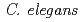 is considered a useful system to investigate the rhythmic phenomena. However, it is difficult to measure the membrane potentials of individual cells, and the mechanisms of rhythm generation and control in pharynx are not well understood. In this study, we propose a simulation-based approach to investigate the mechanisms of rhythm phenomena. To conduct the simulations, we developed a pharyngeal muscle model including 29 cell models, which simulate the activity of each cell as a membrane potential based on the FitzHugh-Nagumo equations. Subsequently, we calculated the EPG by using the outputs of all cell models. As the result, our model successfully generated similar EPG to that observed in a wild type. Furthermore, we could simulate the responses of some mutants such as
is considered a useful system to investigate the rhythmic phenomena. However, it is difficult to measure the membrane potentials of individual cells, and the mechanisms of rhythm generation and control in pharynx are not well understood. In this study, we propose a simulation-based approach to investigate the mechanisms of rhythm phenomena. To conduct the simulations, we developed a pharyngeal muscle model including 29 cell models, which simulate the activity of each cell as a membrane potential based on the FitzHugh-Nagumo equations. Subsequently, we calculated the EPG by using the outputs of all cell models. As the result, our model successfully generated similar EPG to that observed in a wild type. Furthermore, we could simulate the responses of some mutants such as 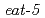 mutant by ablating certain gap junctions virtually so as to correspond to the defects.
mutant by ablating certain gap junctions virtually so as to correspond to the defects.
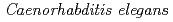
曽 智*; 辻 敏夫*; 鈴木 芳代; 服部 佑哉; 滝口 昇*; 大竹 久夫*
no journal, ,
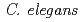 is considered the model system to investigate information processing mechanisms in neural circuits, and some mathematical models have been proposed. However, effects of body dynamics on information acquisition processes were neglected in the previous studies. In this study, we proposed a simulation platform comprising of environmental, neural, and body dynamics models. In our simulation platform, the body is approximated using a multi-joint rigid link model which can produce dynamic body motion commanded by the neural model. As an application example, we analyzed mechanisms of chemotaxis to NaCl. The previous studies revealed that the animal employs pirouette mechanism and weathervane mechanism for the chemotaxis. The two mechanisms respectively require temporal and spatial gradient of NaCl. To discuss the acquisition mechanism of the chemical gradient, we defined environmental model to calculate diffusion of NaCl solution on the agar plate, and approximated input-output characteristics of ASE neurons based on results of calcium imaging. Response of ASE neurons can be calculated by providing head coordinate of the body dynamics model. We also assumed a neural model to convert the responses of ASE neurons to temporal and spatial gradient of NaCl and then to command the body model. We will discuss the results of chemotaxis simulation by comparing to data of actual animals.
is considered the model system to investigate information processing mechanisms in neural circuits, and some mathematical models have been proposed. However, effects of body dynamics on information acquisition processes were neglected in the previous studies. In this study, we proposed a simulation platform comprising of environmental, neural, and body dynamics models. In our simulation platform, the body is approximated using a multi-joint rigid link model which can produce dynamic body motion commanded by the neural model. As an application example, we analyzed mechanisms of chemotaxis to NaCl. The previous studies revealed that the animal employs pirouette mechanism and weathervane mechanism for the chemotaxis. The two mechanisms respectively require temporal and spatial gradient of NaCl. To discuss the acquisition mechanism of the chemical gradient, we defined environmental model to calculate diffusion of NaCl solution on the agar plate, and approximated input-output characteristics of ASE neurons based on results of calcium imaging. Response of ASE neurons can be calculated by providing head coordinate of the body dynamics model. We also assumed a neural model to convert the responses of ASE neurons to temporal and spatial gradient of NaCl and then to command the body model. We will discuss the results of chemotaxis simulation by comparing to data of actual animals.